Reactivity trend on periodic table3/8/2024  
And the farther they are from the righ side of the table, the weaker their electronegativity is, resulting in lower Chemical Reactivity. predict which element differs the most from. predict which element forms the most stable compounds in the +2 oxidation state. 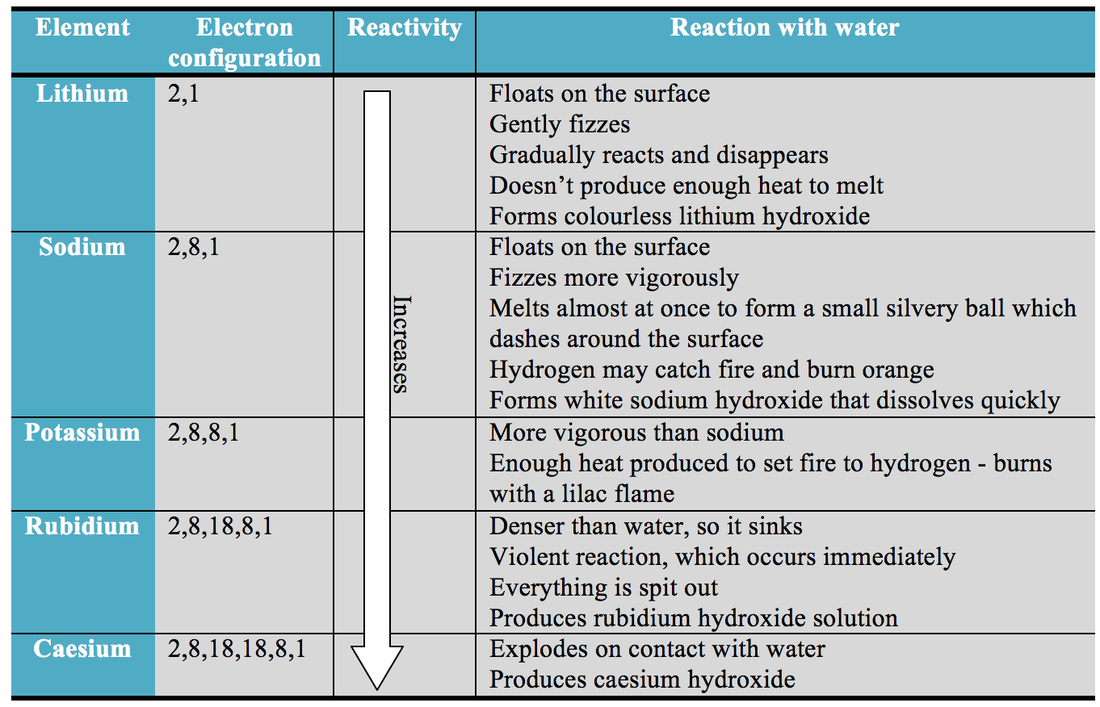
That is why as you go up a group Chemical Reactivity increases because it is easier for elements to gain electrons when they have high electronegativity. Based on the positions of the group 14 elements C, Si, Ge, Sn, and Pb in the periodic table and the general trends outlined in this section, classify these elements as metals, semimetals, or nonmetals. Chemical Reactivity decrease as you go down the groupįor Non-Metals, the farther right-up in the table you go, the higher the electronegativity.Chemical Reactivity increases as you go left to right of the periodic table.And you go from left to right on the periodic table elements have more electrons in their valence shells they have to get rid off which requires an element to have high energies which results into lower Chemical Reactivity. This happens because as you go down a group, it is easier for electrons to be taken or given away, resulting in high Chemical Reactivity. Chemical Reactivity increases as you go down the group.Chemical Reactivity decrease as you go left to right of the periodic table.Īnother factor is the Electronegativity of the element, which determine how badly an element wants to take other electrons from other elements. It can be used as a differentiated activity for the more able students within a group. This could be used to follow up some work on the periodic table where the trends in reactivity in groups 1 and 7 have been identified. One factor is Ionization Energy, Ionization Energy is how easily and element can remove its electrons. Chemistry for the gifted and talented: trends in reactivity in the periodic table. Trends of chemical reactivity in Periodic Table The systematic arrangement of elements in a periodic table discloses certain periodic trends in the properties of elements. A couple factors will determine how likely and element will react to another element.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
